Introduction
The acid-base dissociation constant (pKa) of substances is a physicochemical property of critical importance to pharmaceutical chemistry. Ionization of a drug can impact a myriad of parameters such as solubility and ADME. While several methods exist for pKa determination, multiplexed capillary electrophoresis (CE) is a popular choice that offers chemists advantages such as high throughput capabilities and small sample quantity requirements. Since 2009, Analiza has employed multiplexed CE to acquire pKa data for tens of thousands of client samples; however, as is the case with many established assays, these protocols were optimized for compounds with a small MW (< 500 g/mol). A deep dive of Analiza’s year-to-date data seems to indicate that the pharmaceutical industry has seen a fundamental shift in focus on larger, more complex molecules, and with it, the need to adapt to meet the industry’s needs.
Experimental Protocol
As a CRO, Analiza does not synthesize its own compounds; however, as a partner in the drug discovery pipeline, we recognize the importance of data acquisition and integrity. The misconception can be that assay protocols are standardized and inflexible; while it is true that some processes have limited wiggle room, Analiza has always prided itself on the ability to customize assays that meet the needs of clients. As it pertains to the pKa assay, the following graphic highlights the decision-making process that guides testing protocols:
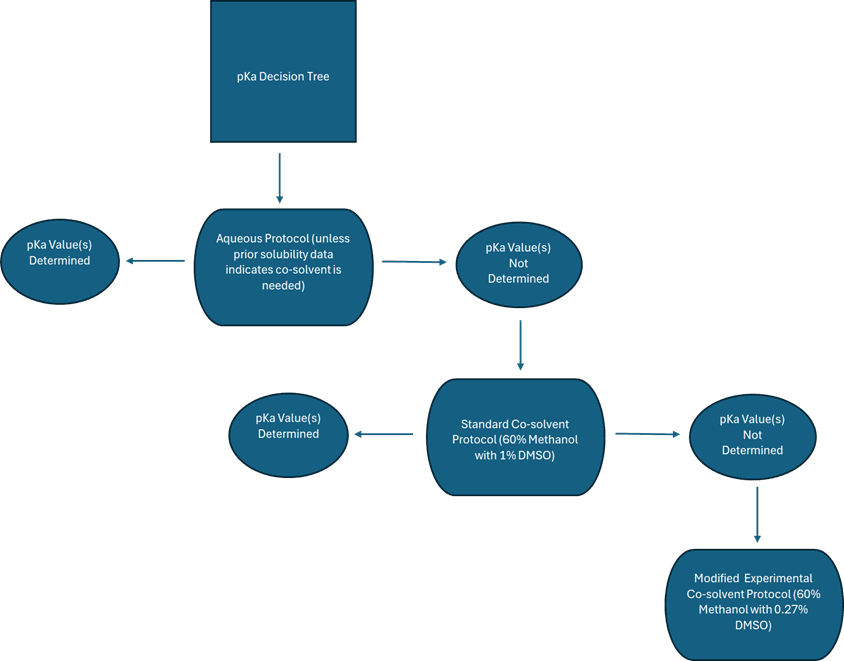
Discussion
From the beginning of 2016 to August 2024, Analiza has run and processed nearly 25,000 pKa samples. A closer look at that data illustrates some interesting trends:
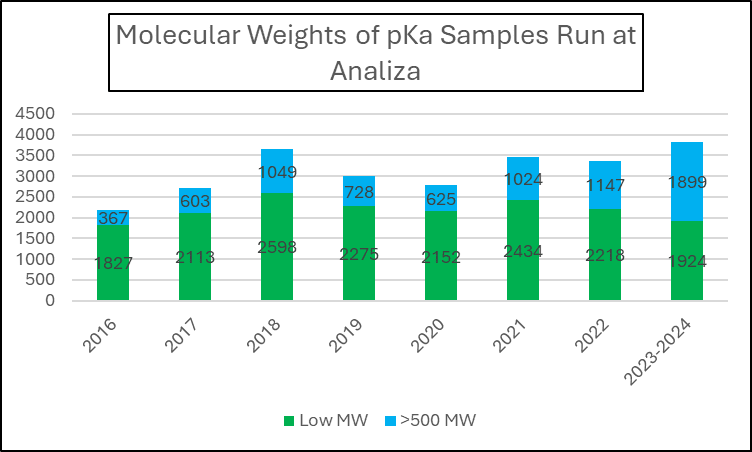
This first chart breaks down the relative molecular weights of tested samples. In 2016, a mere 367 of the 2194 samples had a MW > 500 g/mol (16.7%). Juxtaposed with 2023-2024, 1899 of the 3823 samples met this criterion (49.7%).
However, it is also important to note how this trend potentially impacts assay protocols and data acquisition:

In 2016, 863 of 2194 samples (39.3%) required the addition of a co-solvent (methanol). Compared with the period of January 2023 to August 2024, Analiza ran 3823 pKa samples, and nearly 50% (50.2% to be exact) of these samples required the addition of a co-solvent. Furthermore, for compounds with a MW > 500 g/mol, the likelihood of the co-solvent protocol being employed rose to 60.3%.
The two charts indicate that the complexity of molecules seems to be increasing, which results in more co-solvent runs. While the co-solvent protocol significantly improves compound solubility, several cases of compounds crashing out of solution persist, either compromising the accuracy of reported pKa values or resulting in a lack of data. In cases where “no data” was reported to clients, it is important to state that several factors could contribute to this: poor compound solubility, inadequate absorbance at the measured wavelength (229 nm), the presence of ionization sites which fall outside of the measurable assay range (pH 1.75 – 11.2), and compound instability to name a few.
As the pharmaceutical industry has transitioned from small molecules to larger bRO5 structures such as PROTACs and macrocyles, the need for revised methods has become more evident. The complexity of the molecules reflects the complexity of the measurements and work needed to acquire pKa values: solubility, extreme ionization sites (pH<1.7 or pH>11.2), and the number and proximity of ionization centers all present unique challenges for data acquisition and interpretation.
While not a synthesis lab, Analiza views itself as a vital partner in the drug discovery pipeline. From incorporating high MW commercial compounds (ledipasvir and amiodarone) as assay standards to testing modified co-solvent methods, Analiza is actively working on viable solutions to meet the industry’s needs while delivering the same high quality data clients have come to expect. Initial testing of a modified method has shown potential and promise while maintaining the high-throughput nature and low sample volume requirements of the current protocols; however, it is important to note that more experiments containing experimental compounds and tinkering of the protocol are needed to provide a robust solution to the challenge of acquiring quality high-throughput data for large molecules.
Written By:
Justin Longenberger
Analytical Chemist III


